Dr. Lawrence Broxmeyer, M.D.
© U.S. Library of Congress
All rights reserved
October 23, 2014
Today, the Ebola, and for that matter the Marburg virus have assumed general acceptance. Yet according to David Rasnick, PhD, a previous member to the Presidential AIDS Advisory Panel of South Africa, there is no convincing evidence, and “certainly no confirmatory evidence of human isolation”, in either case. This is ― to be certain ― not only a direct challenge that the “Ebola” is behind the current Ebola outbreak ― but that the virus is pathogenic at all. Nor is Rasnick alone. If the virus called Ebola is not causing the current epidemic, then what is?
The CDC (Centers for Disease Control and Prevention) recently declared:
“Diagnosing Ebola in a person who has been infected for only a few days is difficult, because the early symptoms, such as fever, are nonspecific to Ebola infection and are seen often in patients with more commonly occurring diseases, such as malaria and typhoid fever.”1 Only a sin of omission. then, would explain why anyone or any group would not want to specifically mention the most commonly occurring cause of infectious death in Africa ― tuberculosis ― whose sky-high rates in West Africa make Ebola look like a dropper-full of water squeezed into the Mississippi.
If by October, 2014, Ebola had laid claim to what WHO (the World Health Organization) claimed was well over 4,000 deaths since its February outbreak2 ― certainly this ought to be weighed in the light of the approximately 600,000 Africans slain by TB during the same window of time.3 Furthermore, if by October 17th, 2014 world-wide Ebola mortality stood at 4,811 then surely 81% of these deaths took place in Liberia and Sierra Leone. While Librarian health officials warned4 as early as 2009 that TB was skyrocketing out-of-control ― a mixed scientific coalition from Sierra Leone and Germany cautioned that Sierra Leone’s tuberculosis level was not only the highest in West Africa, but filled with resistant strains of TB and tuberculous Mycobacterium Africanum that had “reached an alarming level”…..raising the question of possible consequences” for a future new TB epidemic.”5 Indeed almost half of all TB cases in the West African Ebola zone are caused by such Mycobacterium africanum ― an unusual, yet just as deadly member of the tubercular family, exclusive to West Africa ― and fast becoming a microbe of great public ― and now global concern. That tubercular M. africanum can and has already caused tuberculosis in the United States is a matter of record.6 Furthermore, there is a body of evidence that africanum requires more sustained contact, even among household members – certainly mirrored in the current outbreak. Meanwhile health officials continue to insist that “casual contact” cannot transmit Ebola ― precisely the same claim they’ve long made with TB.
Surely the CDC is aware that there is not a sign or symptom of Ebola, including its hemorrhagic tendencies that cannot be found in acute disseminated miliary (blood-borne) tuberculosis, once called “galloping consumption” ― the single most feared form of the disease ever. And most likely it is also aware that such tuberculosis has its own viral-like forms, some of which can simulate the Ebola. Such viral TB is generally acknowledged to be TB’s preferred form ― as a survival strategy to storm any inclement conditions the microbe might find itself in.7
Then why did the CDC not mention TB, by name, in their short-list of possibilities that could cause Ebola-like symptoms? If such oversight stopped there it would be unremarkable, but it seems to have been carried over in the very design of the most recent CDC-approved tests to detect Ebola.
A River named Ebola
In September of 1978, about 40 years ago, a team ― including a 27-year-old fresh out of medical school, who was training as a clinical microbiologist at the Institute of Tropical Medicine in Antwerp, Belgium, received a blue thermos from Zaire. It was filled with the two 5ml. clotted blood specimens of an African-based Flemish nun. The Belgium doctor who sent it, Jacques Courteille, practicing in Kinshasa, included a note saying that he was at a complete loss for the nun’s mysterious, yet deadly illness. Also, could the samples be tested for Yellow Fever? This thermos had traveled from Zaire’s capital city of Kinshasa, on a Sabena commercial flight to Belgium ― inside its deliverer’s hand luggage. When the samples were received, Peter Piot, the 27-year-old medical graduate and his colleagues, among other things, placed the blood samples under an electron microscope. To be sure Piot’s interest was virology and a virologist he would soon become, best known for his work on theorizing the ‘viruses’ behind Ebola and AIDS. To this effect he contributed heavily to the voluminous literature that HTLV-1 had a role in AIDS, which it did not. Nevertheless Piot would become a pioneer, and part of the group that included veterinarian Max Myron Essex in trying to define what AIDS was at WHO’s 1985 Conference in Banqui, Africa.

Dr. Peter Piot
Piot on the Ebola: “We saw a gigantic worm like structure ― gigantic by viral standards. It’s a very unusual shape for a virus, only one other virus looked like that and that was the Marburg virus.”8

Figure 2. Piot (on the right), at the Institute of Tropical Medicine, Antwerp, in 1976
But the new “virus” needed a name. Piot relates the interesting tale of how Ebola came to be named as Ebola:
“On that day our team sat together late into the night – we had also had a couple of drinks – discussing the question. We definitely didn’t want to name the new pathogen “Yambuku virus”, because that would have stigmatized the place forever. There was a map hanging on the wall and our American team leader suggested looking for the nearest river and giving the virus its name. It was the Ebola River. So by around three or four in the morning we had found a name. But the map was small and inexact. We only learned later that the nearest river was actually a different one. But Ebola is a nice name, isn’t it?”9
Depends upon how you look at it.

Figure 3. The Ebola River, Circa 1976.
Piot’s specimens proved negative for Yellow Fever and he mentions that the tests for Lassa fever and typhoid were also negative. What, then, could it be? Piot: “To isolate any virus material” small amounts of the blood samples were injected into VERO cells and into mice. Several of these mice subsequently and abruptly expired ― “a sign that a pathogenic virus was probably present in the blood samples that we had used to inoculate them”.10
The fact that the mice died did not mean that it was at the hands of a “pathogenic virus.”
Piot’s boss, Stefaan Pattyn, who Piot admitted “could be a bit of a bully”, supposedly specialized in the study of mycobacteria ― tuberculosis and leprosy, yet seemed unaware of the hemorrhagic consequences of acute TB, nor had he taken the time to use special stains and cultures to detect its viral-like cell-wall-deficient forms. Instead Pattyn followed his current passion, shared by Piot. Pattyn had recently worked in Zaire for six or seven years and exotic viral illnesses were now “right up his alley”.11 So Pattyn’s team likewise never really considered a strain of acute miliary TB or its viral cell-wall-deficient forms in his rule-outs for an acute hemorrhagic or epidemic fever ― among them Mycobacterium tuberculosis and Mycobacterium africanum.
From Past to the Present
The Ebola of its day on steroids, “galloping” acute consumptive tuberculosis could kill in days ― the mere memory of which, just a few generations ago, brought terror to the faces of those who had witnessed and were describing it. To Dubos12 “galloping consumption” was not an isolated, but a frequent diagnosis in the 19th and early 20th centuries. And despite persistent myths to the contrary, in the early phase of any new TB epidemic from a new and virulent strain, tuberculosis manifests itself as an acute disease and only much later as the chronic pulmonary tuberculosis that we know in today’s western world. An example of this can be found in the high mortality during the 1918 influenza pandemic, when African-Americans were brought to fight in France during World War I ― large numbers of them dying from a fast-tracked tubercular “galloping consumption”.
Many often underestimate the speed, contagiousness and ferocity of a TB epidemic. Khomenko’s 1993 study13 should have cemented the notion that the explosive contagiousness of just such Ebola and influenza-like viral forms of tuberculosis are exactly the stuff that previous epidemics and pandemics could have been made of. But it didn’t.
In the US, the CDC and NIH seemed to feel differently, ignoring the historic possibility. There was much the same viral passion, at that time over “Influenza”, when in 1990, a new multi-drug-resistant (MDR) tuberculosis outbreak took place in a large Miami municipal hospital. Soon thereafter, similar outbreaks in three New York City hospitals left many sufferers dying within weeks. By 1992, approximately two years later, drug-resistant tuberculosis had spread to deadly mini-epidemics in seventeen US states, and was reported, not by the American, but the international media, as out of control. Viral forms of swine, avian and human TB can be transmitted from one species to another. So can exotic strains of tuberculosis and tubercular Mycobacterium africanum, imported into the United States through countries such as Liberia. By 1993 the World Health Organization (WHO) had proclaimed tuberculosis a global health emergency.14 That emergency has never been lifted.
Anderson pointed out that such acute, untreated disseminated, “galloping”, blood-dispersed TB could kill in hours or days15 ― its mortality, according to Saleem and Azher even today approaching 100%.16 Ebola itself can take up to a month to kill its victims, said Ben Neuman17, an expert in viruses at Britain’s Reading University ― although there are many cases that also kill in hours or days. Not only were tubercular hemorrhaging and fever both mentioned by Fox18, but hemorrhaging of the serous cavities, the gums, and the nose, into the joints, the skin, and the bowels. Appleman19, in the American Journal of Ophthalmology, considering massive spontaneous hemorrhages into the vitreous, mentions that Axenfeld considered acute tuberculosis an important possibility in the rule-out for bleeding into the eye. Coughing-up blood has always been a well-known scenario for TB. Hemorrhages of significance from the ear secondary to tuberculous otitis media are also on record.20 And the possibility of acute disseminated tuberculosis attacking the bone marrow and through fibrosis causing a partial shutdown of platelets; changing the very morphology of those platelets; as well as interfering with their function ― all combine to create a clear and present hemorrhagic danger. Even today, bone marrow biopsy is at times a valuable diagnostic test for tubercular involvement. In addition Extrapulmonary (outside of the lungs) tuberculosis is the most frequent cause of a prolonged Fever of Unknown Origin (FUO) and has been for a long, long time.21
A fact initially carefully minimized by certain Ebola ‘authorities’ ― and clarified by Feldmann in the New England Journal (October 9th, 2014 issue) is that in the current Ebola outbreak “less than half” of the people infected have visible hemorrhaging.22 This was just enough to prompt some virologists to rethink Ebola’s designation from Ebola Hemorrhagic Fever, to the “Ebola virus Disease”. So much for “hemorrhagic fever.” Yet even then, in a 1978 publication of the Bulletin of the World Health Organization regarding the 1976 Ebola outbreak in Zaire it was admitted that hemorrhaging, although from “multiple sites” was “principally [from] the gastrointestinal tract”.23 But patients with TB spread to the gastrointestinal tract can also have fever, abdominal pain, and can have gastrointestinal/rectal bleeding that patients with Ebola can have.
Mortality Rates of Both Diseases about the Same
Nor do the parallels stop there. In September, as the CDC justifiably warned against nonessential travel to Sierra Leone ― available data from the two Ebola facilities in that country came in with Case Fatality Rates (CFRs) for Ebola that ranged between 50% and 72%. This, although considerably higher than the 37.7% CFR that Sierra Leone’s Ministry of Health was reporting24, averages out to an agency reported fatality rate of 61% ― not much different from the approximately 67% mortality given for the untreated active tuberculosis that currently rages in West Africa and many other places around the world.25
Meanwhile, The World Health Organization’s (WHO’s) latest “Situation Report” summed-up that although the rate of Ebola infections were picking up speed at an alarming rate in West Africa, the fatality rate was 53% overall, ranging from 64% in Guinea to just 39% in Sierra Leone.26
A Flaw in Diagnostic Test Design?
Moreover, the design of present diagnostic tests for Ebola, in certain respects, don’t meet the sniff test. An August 6, 2014 article in the L.A. Times mentioned27 that an unapproved Ebola test-tube diagnostic assay, developed by the U.S. military, was just approved for use in the US under a special emergency-use provision.
Critics claimed that the two PCR systems to be used for Ebola testing in such “emergency situations” were unapproved. But there is more. While an instruction booklet issued by the Food and Drug Administration28 showed impressive results for detecting and thereby being positive for known “Ebola” samples ― it sadly failed in its inadequate selection of those pathogen’s that might be cross-reacting and therefore making for false positive Ebola tests.
The instruction booklet, Version 2, that accompanied the new Ebola assay mentions:
“9.2.2.2 Bacterial Cross-Reactivity: Bacterial cross-reactivity of the EZ1 assay was evaluated by testing purified nucleic acid of bacteria that potentially could be infecting the majority of the population. No cross-reactivity was observed in the human DNA or any of the bacteria tested (Table 51).”29 [Bold print theirs.]
Yes. The only problem being that a glance at Table 51 shows practically every bacteria in existence except for the one subset of pathogens “that potentially could be infecting the majority [of West Africa’s] population” and those pathogens are again ― Mycobacteria tuberculosis and its related Mycobacterium africanum.
Such diversion is no trivial point. As time went by it became obvious that attempts were in the pipeline to link the pathogenesis of Ebola and AIDS, right down to their sexual transmission (a poorly kept secret is that TB can also be sexually transmitted30). So mistakes made during the AIDS probes would have to be avoided with Ebola.
For example, in the past, as the first scientist to propose HIV-virus testing, veterinarian Max Myron Essex knew that tuberculosis and its allied mycobacteria gave a false positive for the HIV virus in his tests in almost 70% of cases. Such cross-reactivity between HIV and tuberculosis was so significant, that it forced Essex31 and his protégé, Congo physician Oscar Kashala, to warn that both the HIV screening test, the enzyme-linked immunosorbent assay (ELISA) and western blot results “should be interpreted with caution when screening individuals with M. tuberculosis or other mycobacterial species.”31 This, of course, automatically meant throwing away HIV serum diagnostics for, according to WHO, at least a third of the people in the known world that WHO (The World Health Organization) has proclaimed presently harbor tubercular infection.
So why then was Mycobacterium tuberculosis noticeably excluded from the CDC’s Table 51 and not included in those pathogens tested for cross-reacting and therefore possibly giving false positive tests for the Ebola? Did the originally panel (Version 1) chosen by government scientists actually include Mycobacterium tuberculosis and related microbes in its design ― only to find that indeed these mycobacteria caused positive tests for Ebola as in the HIV affair? Did they feel that such results might muddy the waters, be too difficult to explain, and subsequently remove them? This is not known.
A group of researchers from Oxford University and the University of Leuven have just determined that HIV is “almost certain” to have begun its spread ― from Kinshasa, now the capital city of the Democratic Republic of Congo.32 Whether this research bears out or doesn’t, Kinshasa itself has long been a hotbed for tuberculosis ― and now Ebola. On top of that, a doctor in rural Liberia, swarmed with Ebola patients, says he’s had extremely good results with HIV treatment ― albeit such treatment was born out of admitted desperation.33 The US NIAID, having gotten wind of this, is carefully looking into the use of some of these HIV-antiretrovirals to control Ebola. This just might work, but will it answer the reason as to why it works? The NIH, for example, has long known, though mechanisms not yet clearly worked out, that HIV treatment suppresses both the tuberculosis and the fowl tuberculosis that are currently the leading causes of infectious death in AIDS. To this effect the NIH, decades ago recruited University laboratories to look into the reason why.34
Therefore are the HIV drugs working against an “Ebola” which is estimated to have killed well over 3,000 African’s so far this year, or the TB that killed 600,000 Africans in that same window? There still remains much work ahead to determine this. Antiretrovirals have major side effects.
The Ebola virus (also referred to as Ebola Hemorrhagic Fever) is often compared to the Marburg virus. So we have this, written in July, 2005, regarding that outbreak:
“Angola is in the grip of the world’s worst ever outbreak of the Marburg virus. According to the World Health Organization (WHO), as of 5 April, 156 of the 181 people reported infected have died. The Ebola-like virus causes a fever that, in fatal cases, is usually accompanied by severe internal bleeding and shock. There is no vaccine or medical treatment and up to 80 per cent of infected people die within three to seven days. Three-quarters of those affected are children under five. Diagnosing an infection with the Marburg virus can be difficult as its initial symptoms are similar to those of malaria or tuberculosis. They include diarrhea, stomach pains, nausea, and vomiting and severe chest pains.”35
The heavy mortality and morbidity under the age of five with Marburg brings to mind specifically a tubercular involvement, in which most children are affected also in the same age group. What has been called ‘The Golden Age of Resistance’ against TB mortality has always been, for unknown reasons, ages six thru fifteen.In this same vein, during the Zaire Ebola outbreak of 1976, women 15-29 years of age had the highest incidence of that disease.36
“We saw a gigantic worm like-structure ― gigantic by viral standards. It’s a very unusual shape for a virus” – Peter Piot.

Figure 4. The Serpentine Form of the Ebola Virus. Magnification: approximately x60,000.
Micrograph from F. A. Murphy, University of Texas Medical Branch, Galveston, Texas. Courtesy: CDC Dr. Frederick A. Murphy
To some it might be considered “worm-like”, to others serpentine.
TB OR EBOLA?

Figures 5A through 5G. Which of the serpentine, worm-like forms above is the Ebola virus? The correct answer is Figure 5 F. Figures 5A, 5B and 5C are L-Forms (Or Cell-Wall-Deficient Forms) of TB Under the Electron Microscope. (Michailova, L et al. Morphological variability and cell wall deficiency in ‘heteroresistant’ strains. Internat Journ of Tuberc and Lung Dis, 9:,8, Aug 2005:.907-914:,911). Figure 5D. Worm-like lethal tubercular cords from an atypical TB under the Electron Microscope. (Julián E, Roldán, M Journ. Of Bact Apr. 2010 p.1751-1760. P.1756). Such virulent cords are also represented in Figure 5E (Darzins, E. The Bacteriology of Tuberculosis Minneapolis. 1958. 488pps. p296) and Figure 5G.
Both the Ebola virus and the Marburg virus are called Filoviruses because they form filamentous infectious viral particles. Filoviruses, however, are not alone. Mycobacterium. tuberculosis, which commonly lodges in and multiplies in the white cell defenders of our body (called macrophages) ― also become filamentous once inside a macrophage.37 (See figures 6,7)
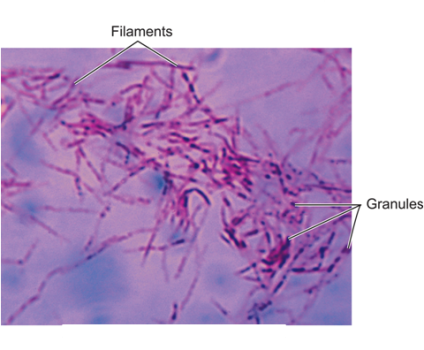
Figure 6. Filamentous Cell-Wall-Deficient Forms of Mycobacterium tuberculosis.

Figure 7. Filamentous Forms of the Ebola. Magnification: approximately x40,000.
Micrograph from F. A. Murphy, University of Texas Medical Branch, Galveston, Texas.
According to the WHO, close contact with the bodily fluids of people infected with Ebola, for example in hospitals or at burials, has in the past increased the risk of infection. Health workers have been advised to wear a mask and gloves.38 Yet physicians all masked and gloved up have contracted Ebola and Marburg.
WHO’s statement is misleading. As far back as 1995 the ability of Ebola to aerosolize or spread through airborne transmission was reported, studied and confirmed.39,40 Ebola, is a communicable airborne infection, just like tuberculosis.
Tubercular Monkeys, Apes and Fruit Bats
“Ebola virus has been found in African monkeys, chimps and other nonhuman primates such as fruit bats in Africa. A milder strain of Ebola has been discovered in monkeys and pigs in the Philippines.”41 Interestingly Infection by M. africanum has also been reported from chimpanzees and African green monkeys. And in 1970-71 the CDC estimated that tubercular infection in individuals in contact with these and other non-human primates was 60 to 100 times that of the population at large (Richardson, 1987). Both African moneys and great apes are susceptible to TB.42 Early studies suggested that the new strain of Ebola had emerged in West Africa, but according to epidemiologist Fabian Leendertz, a disease ecologist at the Robert Koch Institute in Berlin, who led the large team of scientists to Guinea, it is likely the virus in Guinea is closely related to the one known as the Zaire Ebola Virus, identified more than 10 years ago in the Democratic Republic of the Congo.43
It is felt that the new strain probably arrived in West Africa via infected straw-colored fruit bats from another part of Africa and seems to be related to that Zaire Ebola virus identified more than 10 years ago in the Congo. These bats migrate across long distances and are commonly found in giant colonies near cities and in forests. Fruit bats, however, are widely eaten in rural West Africa – supposedly as a delicacy.
But the same fruit bats can carry mycobacteria from the mycobacterial tuberculosis complex.44,45,46
Previous Ebola outbreaks saw catastrophic death rates in gorilla and chimpanzee populations, which led some scientists to think that they were the ones responsible for the disease spreading. Old World monkeys are very susceptible to human and cattle tuberculosis. Unlike humans, monkeys have no natural resistance to the disease. When they do catch it from a human, it usually spreads very fast and fatally in their bodies (as in acute miliary tuberculosis) and to areas other than their lungs. During their illness, they can spread the disease to anyone who comes in contact with them or their waste.
The current Ebola virus began in West Africa, a hotbed of tuberculosis. (See Figure 8)

Figure 8: Map showing the true severity of the tuberculosis problem; in 2006-2008 – the last period before WHO’s TB statistics dived as a result of including many TB cases into the wastebasket of “AIDS-defining illness”. Note that the darker-colored regions are mostly in Africa. (Source: WHO)
Another map recently put out by WHO lumps Ebola deaths in West Africa under three puzzling categories “probable, confirmed, and suspected”.47
So what is the take-home message here? At best that “Ebola-like” does not always mean Ebola; at worst that Ebola does not mean Ebola.
REFERENCES:
1. CDC. Ebola Diagnosis. September 2014. http://www.cdc.gov/vhf/ebola/diagnosis/
2. World Health Organization. WHO: Ebola Response Roadmap Update. Oct. 17, 2014. 4pp. p. 1.
http://apps.who.int/iris/bitstream/10665/136645/1/roadmapupdate17Oct14_eng.pdf
3. Ball, James. Concerned about Ebola? You’re worrying about the wrong disease, The Guardian. 2014, August.
http://www.theguardian.com/commentisfree/2014/aug/05/ebola-worrying-disease
4. LIBERIA: Tuberculosis on the rise, IRIN News. 4 February, 2009
http://www.irinnews.org/report/82747/liberia-tuberculosis-on-the-rise.
5. Homolka et al. High genetic diversity among Mycobacterium tuberculosis complex strains from Sierra Leone.
BioMed Central. 25 June 2008 p.1-8 p.1. http://www.biomedcentral.com/1471-2180/8/103.
6. Desmond, E, Ahmed AT, Probert WS et al. Mycobacterium africanum cases, California, Emerg Infec Dis 2004 May; 10(5):921-923 p.1. http://wwwnc.cdc.gov/eid/article/10/5/pdfs/03-0016.pdf
7. Mattman, LH. Cell Wall Deficient Forms ― Stealth Pathogens 3rd Edition CRC Press Boca Raton 2001 416pp p. 189.
8. Brown, R. The virus detective who discovered Ebola in 1976. BBC News Magazine. http://www.bbc.com/news/magazine-28262541
9. Von Bredow R, Hackenbroch V. Interview with Dr. Peter Piot: ‘In 1976 I discovered Ebola – now I fear an unimaginable tradgedy.” Der Spiegel through The Observer. 2014, Oct 5. http://www.theguardian.com/world/2014/oct/04/ebola-zaire-peter-piot-outbreak
10. Piot, P. Part one: A virologist’s tale of Africa’s first encounter with Ebola. Science. American Association for the Advancement of Science (AAAS). 2014, Aug. http://news.sciencemag.org/africa/2014/08/part-one-virologists-tale-africas-first-encounter-ebola
11. Ibid.
12. Dubos R, Dubos J. The White Plague: Tuberculosis, Man, and Society, Rutgers University Press, 1987, p. 205.
13. Khomenko AG, Muratov VV. Epidemiological risks of tuberculous infection foci in patients discharging L forms of Mycobacterium tuberculosis. Problemy Tuberkuleza i Bolezni Legkikh, 2,2–5 1993.
14. Reuters. WHO Calls Tuberculosis a Global Emergency. Health Statistics. Los Angeles Times, 24 April 1993, http://articles.latimes.com/1993-04-24/news/mn-26683_1_global-emergency
15. Anderson, M. Clinical Lectures On the Curability of Attacks of Tubercular Peritonitis and Acute Tuberculosis (Galloping Consumption). James Maclehose Publisher. Glascow. 56p. 1877.
16. Saleem A and Azher M. The Next Pandemic – Tuberculosis: The Oldest Disease of Mankind Rising One More Time. British Journal of Medical Practitioners, June 2013; 6 (1):a615.
17. Kelland K. Ebola mortality rate expected to rise as outbreak runs its deadly course. Reuters. Aug 5, 2014. http://news.yahoo.com/ebola-mortality-rate-expected-rise-outbreak-runs-deadly-192146284.html
18. Fox,W. A treatise on Diseases of the Lungs and Plueura. Coupland, S, editor. J.&A. Churchill. London. 1891 1200 pp.
19. Appleman LF. Massive spontaneous hemorrhages into the vitreous Am Journ of Ophth. Vol. 1 No. 1, January, 1918. P24-28.
20. Burnett CH. A Text-book on Diseases of the Ear, Nose and Throat. J.B. Lippincott. 1901. 716pp.
21. Kasper DL, Braunwald E, Fauci AS, Hauser SL, Longo DL, Jameson JL, Casademont J. Harrison’s Principles of Internal Medicine. 16th Edition. 2004. McGraw-Hill Professional Publishing. 2680 pp.
22. Feldmann H, Ebola ― A Growing Threat? N Engl J. Med 371;15. p1375-1378 p.1374; October 9, 2014.
23. Report of an International Commission. Ebola haemorrhagic fever in Zaire, 1976. Bulletin of the World Health Organization, 56(2): 271-293 p.271. 1978
24. Conton, P., “Sierra Leone News: Ebola – what is the true Case Fatality Rate CFR?, September 2nd, 2014, http://thethirdway.forumchitchat.com/post?id=7062282&goto=nextnewest
25. Saleem and Azher; op. cit.
26. Conton, op.cit.
27. Morin, M. FDA authorizes Ebola test; vaccine will probably take until 2015. Aug 6, 2014. http://www.latimes.com/science/sciencenow/la-sci-sn-ebola-vaccine-20140806-story.html.
28. JPM-MSC. Ebola Zaire [EZ1] rTR-PCR [TaqMan®] Assay on ABI® 7500 Fast Dx. LightCycler®, and JBAIDS. Instruction Booklet Version 2.0 14th of August 2014. Manufactured by the Naval Medical Research Center for The U.S. Department of
Defense.<a href=”http://www.fda.gov/downloads/MedicalDevices/Safety/EmergencySituations/UCM408334.pdf” target=”_blank”>http://www.fda.gov/downloads/MedicalDevices/Safety/EmergencySituations/UCM408334.pdf
29. Ibid., p.53 Table 51, pp. 69-70.
30. Broxmeyer, L. AIDS: What the Discoverers of HIV Have Never Admitted: Latest Edition. 2014, July. 142 pp http://www.amazon.com/AIDS-Discoverers-Admitted-Latest-Edition/dp/1495457044/ref=sr_1_fkmr0_3?ie=UTF8&qid=1412347161&sr=8-3-fkmr0&keywords=Lawrence+Broxmeyer+AIDS%3A+What+the+discovers+of+HIV+have+never+admitted.
31. Kashala O, Marlink R, llunga M, Diese M, Gormus B, Xu K, Mukeeba P, Kasongo K, Essex M. Infection with human immunodeficiency virus type 1 (HIV-1) and human T cell lymphotropic viruses among leprosy patients and contacts: correlation between HIV-1 cross-reactivity and antibodies to lipoarabinomannan. J Infect Dis. 1994 Feb;169(2):296-304.
32. Schlanger, Z., Researchers Pinpoint Origin of HIV Pandemic Newsweek, October 2nd 2014. http://www.msn.com/en-us/news/world/researchers-pinpoint-origin-of-hiv-pandemic/ar-BB740Xi
33. Izadi, E., A Liberian doctor is using HIV drugs to treat Ebola victims. The NIH is intrigued, The Washington Post, October 2, 2014. http://www.washingtonpost.com/news/to-your-health/wp/2014/10/02/a-liberian-doctor-is-using-hiv-drugs-to-treat-ebola-victims-the-nih-is-intrigued/
34. Broxmeyer, op. cit., pp.6-17
35. Shetty, P. Worst ever outbreak of Marburg virus hits Angola, SciDevNet, April 7, 2005. http://www.scidev.net/global/health/news/worst-ever-outbreak-of-marburg-virus-hits-angola.html?from=related%20articles&stay=full
36. Dubos and Dubos, op. cit.
37. Chauhan A, Madiraju MV, Fol M, Lofton H, Maloney E et al. Mycobacterium tuberculosis Cells Growing in Macrophages Are Filamentous and Deficient in FtsZ Rings J Bacteriol. Mar 2006; 188(5): 1856–1865.
38. WHO: “Ebola virus disease”, Fact sheet no. 103, updated September 2014, http://www.who.int/mediacentre/factsheets/fs103/en/
39. Johnson E, Jaax N, White J, Jahrling P. Lethal experimental infections of rhesus monkeys by aerosolized Ebola virus. Int J Exp Pathol. 1995 Aug; 76(4):227-36.
40. Jaax N, Jahring P, Geisbert T, Geisbert J, Steele K, McKee K et al. Transmission of Ebola virus (Zaire strain) to uninfected control monkeys in a biocontainment laboratory. Lancet. 1995 Dec 23-30; 346(8991-8992): 1669-71.
41. The Mayo Clinic Diseases and Conditions. Ebola virus and Marburg virus. Aug 6, 2014. http://www.mayoclinic.org/diseases
42. Kaufmann AF, Moulthrop JI, Moore RM: A perspective of simian tuberculosis in the United States—1972. J Med Primatol 4:278–286, 1975.
43. Vidal, J., “Ebola: research team says migrating fruit bats responsible for outbreak”, The Observer, 24 August 2014. http://tinyurl.com/lzdgqry
44. Scott, H.H., “Report on the deaths occurring in the Society’s gardens during 1925”, Proc. Zool. Soc. London. 1926; 96:231-244
45. Griffith, A.S., “Tuberculosis in Captive Wild Animals”, J. Hyg. (Lond.) 1928 Nov; 28(2):198-218.
46. Hamerton, A.E., “Report on the deaths occurring in the Societies gardens during 1930.” Proc. Zool Soc. Lond. 1931: 101:527-555).
47. WHO, “WHO: Ebola Response Roadmap Update. 10 October 2014 http://apps.who.int/iris/bitstream/10665/136161/1/roadmapupdate10Oct14_eng.pdf?ua=1.
Dr. Lawrence Broxmeyer, MD
© U.S. Library of Congress
All rights reserved
October, 2014
SEE ALSO::
Broxmeyer, L. AIDS: What the Discoverers of HIV Have Never Admitted: Latest Edition. 2014, July. 142 pp.
Pennsylvania internist/researcher Lawrence Broxmeyer, MD, was on staff at N.Y. affiliates of Downstate, Cornell, and NYU for 14 years. He was lead author and originator of a novel way to kill AIDS mycobacteria. (Journal of Infectious Diseases, 11/15/2002}. His ideas on phagotherapy are still in use today. He appeared in Patho-Biotechnology, by Landes. His peer-reviewed articles are on PubMed. His books include “Parkinson’s—Another Look”, “Autism: An ancient Foe” and “AIDS: What the Discoverers of HIV Have Never Admitted.” SEE: AIDS: What the Discoverers of HIV Have Never Admitted: Latest Edition








